 The electrons are not moving along the paths of the curved arrows. They represent imaginary electron movement between the resonance forms. Curved arrows are often used to make it easier to understand the relationship between the two (or more) structures (Figure 5.22). Resonance is a graphical tool to approximate the ‘real’ distribution of electron density in the molecule. When represented in these ways it becomes easy for students to lose track of how many electrons (lone pairs and π bonds) are being shown.įigure 5.21 – Alternate Representations of Resonance Structures. 
These are common in biochemistry but are heavily discouraged in this text. Sometimes this is represented using dotted bonds, or a circle in the case of aromatic compounds (Figure 5.21). This is because they imply, together, that these bonds are not double bonds, not single bonds, but something in between. However, when considered together they give a much more accurate picture than any on its own. Each way of drawing the molecule is an equally acceptable approximation, but none of them, by itself, is an accurate picture of the molecule. Which way is correct? There are two answers to this question: ‘all’ and ‘none’. The structures are related to each other using the double headed arrow for resonance (Table 5.1)įigure 5.20 – Examples of Resonance Structures. For example, there are two ways to draw the double bonds in 1,2-dimethylbenzene ( o-xylene), and three different ways of representing N,N-dimethylformamide. These are referred to as resonance structures (Figure 5.20). There are several things that should be checked before and after drawing the resonance forms.Some molecules can be represented as different Lewis or line-angle structures that have the same placement of atoms but a different placement of electrons. The reader must know the flow of the electrons. Make sure the arrows are clear including the single and half headed arrow. The tail of the arrow begins at the electron source and the head points to where the electron will be. # of valence electrons- (#non bonding electrons + 1/2 #bonding electrons)Ĭurved arrow notation is used in showing the placement of electrons between atoms. Assigning formal charges to an atom is very useful in resonance forms.įormal charge is calculated using this format: An atom with many electrons will have a negative charge. Atoms that are missing one or more electrons will have a positive charge. This is why formal charges are very important. 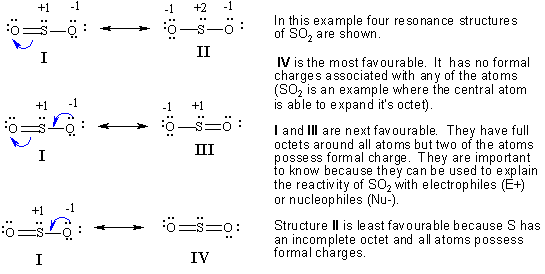
Remember, the best resonance structure is the one with the least formal charge. These structures used curved arrow notation to show the movement of the electrons in one resonance form to the next.įormal charges are used in Chemistry to determine the location of a charge in a molecule and determine how good of a Lewis structure it will be. They are drawn with a double-headed arrow between them to show the actual structure is somewhere between the resonance structures. The more resonance forms a molecule has makes the molecule more stable. The better ones have minimal formal charges, negative formal charges are the most electronegative atoms, and bond is maximized in the structure. Not all resonance structures are equal there are some that are better than others. Resonance structures are a better depiction of a Lewis dot structure because they clearly show bonding in molecules. Resonance forms differ only in arrangement of electrons. Isomers have different arrangement of both atoms and electrons. Resonance structures are not in equilibrium with each other. First resonance structures are not real, they just show possible structures for a compound. There are some basic principle on the resonance theory. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
